Presence of DTCs in bone marrow (BM) predicts reduced survival in early BrCa. The present study explores the use of DTCs to identify adjuvant insufficiently treated patients (pts) to be offered secondary adj treatment (Tx), and as a surrogate marker for Tx response. In this prospective trial, 1121 early BrCa pts were enrolled after completion of 6 cycles of adjuvant FEC chemotherapy from Oct 2003-08. BM aspiration was performed 8-12 weeks after FEC (BM1), followed by a second BM aspiration 6 months (mo) later (BM2). Presence of DTCs in BM was determined by immunocytochemistry using pan-cytokeratin mAbs. If ≥1 DTCs were present at BM2, 6 cycles (mean 5.8; range 3-6) of docetaxel (D)(100 mg/m2, 3qw) was administered, followed by DTC analysis 1 mo and 13 mo after last D infusion (post-Tx). The primary endpoint was disease free survival (DFS). Log-rank statistics and Cox proportional-hazard model were used to compare outcome in pts treated with D with no DTCs after Tx to pts with DTC persistence, and in all pts according to primary tumor factors and DTC status. Of 1067 pts with available FU information (median 80.6 mo; end of FU Nov 2012) and a DTC result at BM2, 8.5% were DTC pos at BM1 and 7.2% at BM2. DTC pos pts at BM2 had higher pT- and pN-stage compared to BM2 neg pts (p=0.035 and p=0.098). Of 72 D-treated pts analyzed for DTCs after Tx, 15 (21%) had persistent DTCs. Pts with remaining DTCs had markedly reduced DFS (47% with relapse) compared to pts with no DTCs post-Tx (HR 6.9, 95% CI 2.2-21.7).
Only 8.8% of D-treated pts with no DTCs post-Tx relapsed, compared to 12.7% of pts in the favorable group with no DTCs both at BM1 and BM2 (p=0.378, log rank). Separate analyses of pts with DTC neg status at BM2, showed that only ER neg pts with DTC pos status at BM1 had reduced survival, compared to those with no DTCs at BM1 (p=0.029). The results of the first part of the study show that DTC status detects high-risk pts after FEC chemotherapy and DTC monitoring status after secondary Tx with D correlated strongly with survival. The results indicate that D contributes to improved prognosis for high-risk pts and emphasize the potential for DTC analysis as a surrogate marker for treatment effect in adjuvant BrCa.
Currently, additional primary tumor analyses (including PAM50) and characterization of the DTCs are performed, aiming at further exploration of the benefit of DTC monitoring and how to best identify those patients that gain most benefit from secondary adjuvant chemotherapy.
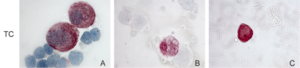
Stained tumor cells (in red) from bone marrow. Synnestvedt et al, BREA 2013.
Selected paper:
PI: Bjørn Naume, Oslo University Hospital. Email: bna(at)ous-hf.no.
















