Background
Tamoxifen (tam) is still an extensively used Estrogen Receptor (ER) antagonist. It is the drug of choice in endocrine adjuvant therapy for pre-menopausal patients and in concert with aromatase inhibitors also for postmenopausal patients. This pro-drug is metabolized by liver enzymes to various active metabolites with serum levels of more than 10-fold inter-individual difference. Controversies exist whether the genetic polymorphisms of the CYP2D6-enzyme or serum levels of active metabolites can be used to evaluate ER inhibition and its effect on survival. Preliminary results from our group suggest that metabolite-guided therapy is the most feasible approach to avoid under-treatment of breast cancer patients receiving long-term endocrine tam treatment.
Design and methods
We have developed a highly sensitive High Pressure Liquid Chromatography-Tandem Mass Spectrometry (LC-MS/MS) method, which is suitable for therapeutic drug monitoring (TDM) also during low tam dose studies. This method can also distinguish between the inactive and active isomers of endoxifen, which is the most active ER-blocking metabolite of tamoxifen (Figure 1). In collaboration with the Oslo Breast Cancer Research Group (OSBREAC), we have recently determined the metabolite profiles in 106 breast cancer patients from the OSLO-1 study in a retrospective observational study. Here, we observed a prognostic relevance of TDM. Importantly, the PBCB-serum samples will enable us to validate this observation prospectively. In the PBCB material and other studies within the network collaboration. TDM may well turn out to be a paradigmatic shift in endocrine tam treatment of ER-positive breast cancer in the future. Importantly, the distance from “bench to bed” in this study is very short due to a simple and robust hypothesis, feasible method and 30 years of experience with tam in the clinical setting. This project is included in Thomas Helland´s PhD project funded by Helse Vest in 2015.
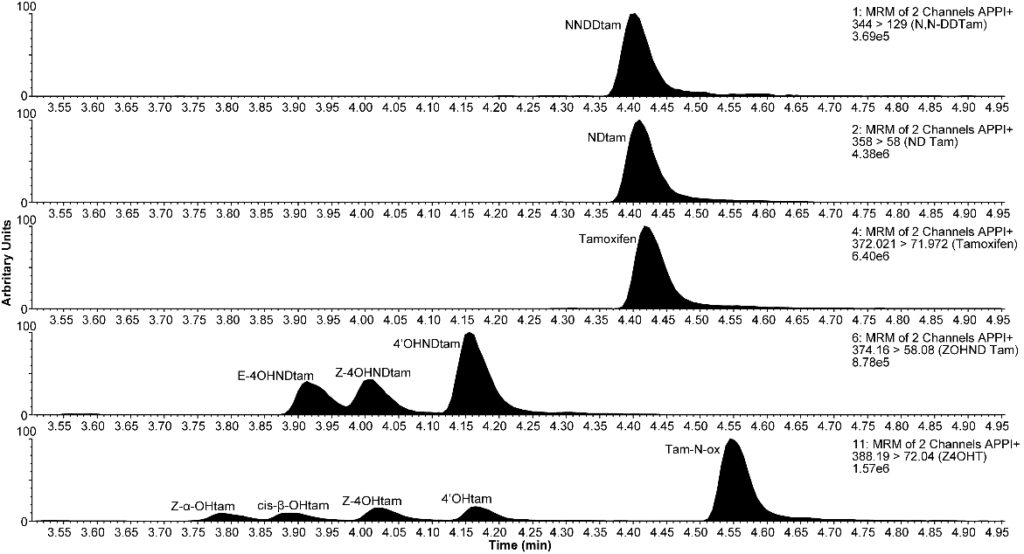
Figure 1: Spectrum of tamoxifen metabolites i serum determined by LC-MS-MS.
PI: Gunnar Mellgren, Haukeland University Hospital, gunnar.mellgren(a)uib.no.
















