SECRET is an EU-funded Marie Skłodowska-Curie Innovative Training Network for early stage researchers (ESRs). The overall objective of this network is to understand the mutual regulation of the secretory and cell signaling pathways in order to identify novel diagnostic or therapeutic strategies for triple negative breast cancer (TNBC). This network has 11 beneficiaries and 7 partners, from both academia and industry. Two Norwegian research groups, headed by Prof Gunhild M. Mælandsmo/senior scientist Lina Prasmickaite from the Institute of Cancer Research (ICR) at Oslo University Hospital and Prof Hesso Farhan from the Institute of Basic Medical Science (IMB) at University of Oslo, are members of this network and will supervise the ESRs in the following projects.
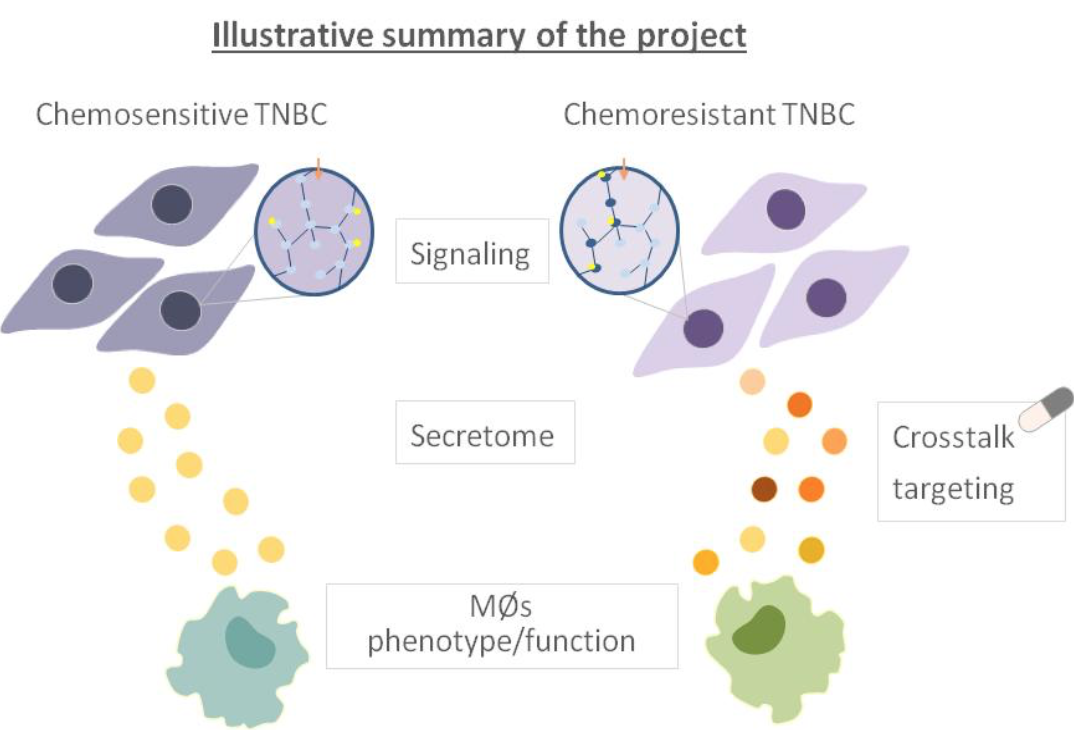
Secretome mediated tumor-macrophage interactions and treatment resistance
Supervisors: Lina Prasmickaite and Gunhild M. Mælandsmo
PhD student: Eleni Skourti
Treatment options for TNBC are limited to chemotherapy, however, the quickly developing chemoresistance is a big challenge and therefore there is a need for better treatment alternatives. In this PhD project we investigate how chemoresistant TNBC interacts with macrophages (MØs), one of the most abundant immune cells in the tumor microenvironment. Specifically, we focus on the role of the secreted factors, known as secretome, in this crosstalk. Thus, by using chemosensitive and chemoresistant TNBC cell lines and patient-derived xenograft (PDX) mouse models we will investigate the composition of the respective secretomes, the regulating signaling pathways, the secretome’s influence on macrophages and finally the therapeutic potential of targeting the resistance-associated determinants. We anticipate that this project may reveal novel targets for therapeutic intervention to improve response to chemotherapy in resistant TNBC.
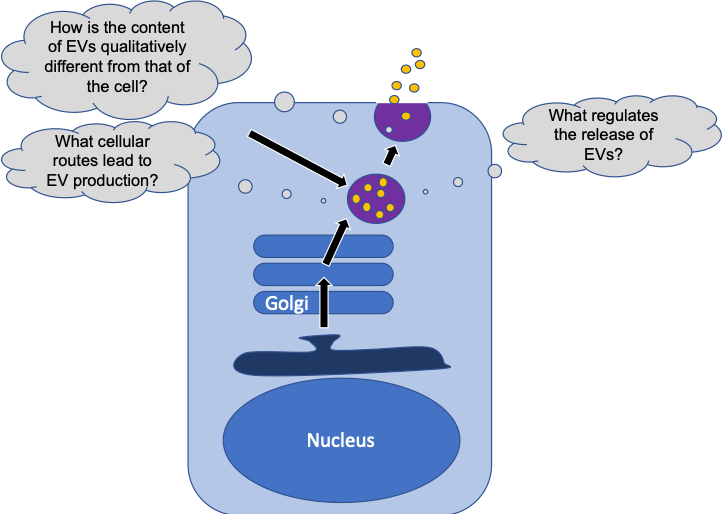
Regulation of Extracellular Vesicle Biogenesis and its Role in Breast Cancer Metastasis
Supervisor: Hesso Farhan
Co-Supervisor: Gunhild M. Mælandsmo
PhD student: Renata Hajdu
One way for cancer cells to communicate with each other is to release small vesicles. These so called extracellular vesicles (EVs) are known to carry many types of proteins, which they can deliver to neighboring cancer cells to modulate their ability to migrate and invade the surrounding tissue. EVs not only help cells communicate with their immediate neighbors, but can also travel to distant organs and prepare a niche that is susceptible to be colonized by cancer cells. Thus, EVs are important players that regulate many aspects relevant for metastatic spreading of cancer cells, making them an important topic for investigation. It remains less well understood how the EVs select their content (cargo) and whether and how proteins inside EVs are qualitatively different from the donor cell that has produced them. Our preliminary data suggest that cargo in EVs is cleaved by proteases. We will investigate the functional significance as well as the cellular location of this cleavage event. This will be achieved using microscopy-based imaging of living breast cancer cells. Furthermore, we will identify triggers and regulators for how EVs select and sort their cargo.


SECRET has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 859962. The project has 11 beneficiaries and 7 partners from academia and industry.

















